Any content you receive is for information purposes only. Always conduct your own research.
*Sponsored
Market Crux Just Announced (CVKD) Will Be Topping Our Watchlist
Tomorrow Morning—Monday, March 02, 2026
Don't Miss The Next Breakout—Get Real-Time Alerts Sent Directly
To Your Phone. Up To 10X Faster Than Email.
(CVKD) Comes Backed By 7 Must See Potential Catalysts
Full Coverage Will Be Starting Early
Pull Up (CVKD) Before Tomorrow Morning…
March 01, 2026
Pull It Up Tonight | See Why (Nasdaq: CVKD) Just Hit Monday's Watchlist Dear Reader, At Market Crux, we live for the moments when a quiet name suddenly starts showing up everywhere—and the screen tells you it's no longer under the radar. In the race to reduce dangerous clots without increasing bleeding risk, one emerging player has been stacking real developments that could influence how this category evolves next. And in a $40B global market where any significant innovation has been limited for decades, the names pushing into underserved corners don't stay quiet for long. That company is Cadrenal Therapeutics, Inc. (Nasdaq: CVKD). And it's just one of the reasons why (CVKD) will be topping our watchlist tomorrow morning—Monday, March 02, 2026. With its expanded clinical assets, operational milestones, and broader pipeline footprint, (CVKD) has moved firmly into our watchlist focus. What caught our attention is the technical setup of (CVKD). (CVKD) has less than 2M shares listed as available to the public. When a company has a small float like this, the potential exists for big moves if demand begins to shift. We have already seen what this can mean on the chart as (CVKD) just made an approximate 73% move inside of 10 days from around $4.91 on February 17 to $8.51 on Friday, February 27, 2026. Analyst Target Suggests Over 450% Upside Potential
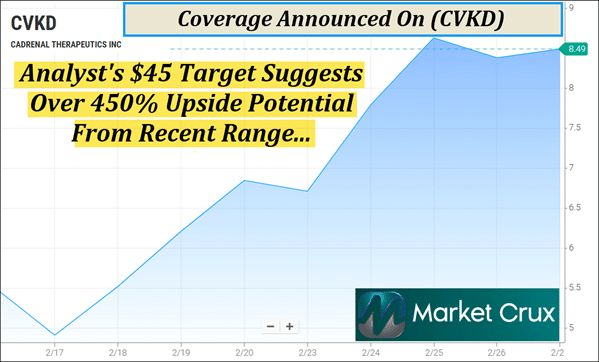
The expansion of the pipeline has been followed by bullish commentary from Noble Capital Markets and Zacks Small Cap Research. Noble Capital Markets: Senior Analyst Robert LeBoyer recently reiterated a Bullish rating with a $45 target on (CVKD). From its recent $8 range, this suggests a potential upside of over 450%. LeBoyer specifically cited the $3B+ peak annual revenue potential of the targeted indications. But here's the key point: the attention isn't showing up from a single headline or a one-off catalyst—it's showing up because of what the company has been building underneath the surface. And the latest late-February updates put a fresh exclamation point on that. Across three recent releases, (CVKD) kept the spotlight on its 12-LOX inhibitor program in Heparin-Induced Thrombocytopenia (HIT) and drew additional analyst attention from Zacks Small Cap Research. On Feb. 20, 2026, Zacks Small Cap Research analyst David Bautz, PhD published coverage focused on the company's acquisition of the 12-LOX inhibitor asset for HIT. In that same coverage, Bautz valued (CVKD) at $25.00/share, which—suggests over 200% upside potential from recent levels. On Feb. 24, (CVKD) reported Phase 2 results for CAD-1005 in HIT. The company highlighted a >25% absolute reduction in thrombotic events versus placebo on top of standard anticoagulant therapy. (CVKD) also noted there was no difference in platelet count recovery and said an End-of-Phase 2 meeting is planned for March 2026. On Feb. 25, a follow-on update reiterated the Phase 2 takeaways. That update was again framed through Zacks coverage, reinforcing the view that the data support continued clinical advancement of CAD-1005. Now let's zoom out—because when a small name pairs this kind of clinical momentum with a broader platform, it helps explain why (CVKD) is starting to look bigger than a single headline. Targeting Unmet Cardiovascular Needs
Cadrenal Therapeutics, Inc. (Nasdaq: CVKD) is a late-stage biopharmaceutical company focused on addressing unmet needs in the anticoagulation and thrombosis space. While the broader market is saturated with therapies, many patient populations remain underserved due to high bleeding risks or unpredictable metabolism. (CVKD) is specifically targeting these gaps with a platform that spans chronic care, acute procedural risk, and immune-mediated thrombosis. The company's primary focus has long been tecarfarin, a next-generation vitamin K antagonist designed for patients with End-Stage Kidney Disease (ESKD) and atrial fibrillation. These patients often cannot use modern therapies like DOACs because they are primarily eliminated by the kidneys. By developing a therapy that is metabolized outside the major hepatic pathways, (CVKD) aims to provide more stable and predictable anticoagulation for those who need it most. In late 2025, (CVKD) transformed from a single-asset company into a multi-program clinical powerhouse. The company recently acquired first-in-class assets that address acute care and rare immune-driven clotting conditions. This expansion has positioned (CVKD) as a versatile player in cardiovascular care, targeting conditions where current therapeutic options often fall short. A Pipeline With Range
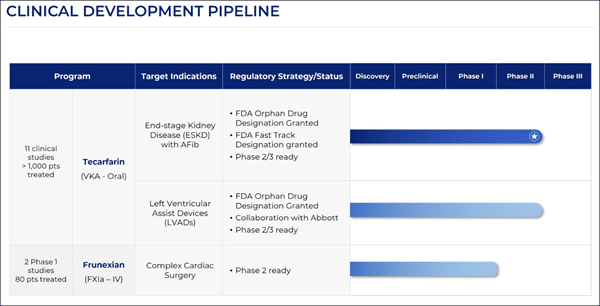
The core thesis for (CVKD) rests on its diversified pipeline, which now features three distinct late-stage programs. The VLX-1005 Breakthrough:
On December 11, 2025, (CVKD) announced the acquisition of VLX-1005, a first-in-class Phase 2 12-LOX inhibitor. This candidate targets Heparin-Induced Thrombocytopenia (HIT), a serious immune-mediated complication of heparin therapy that can lead to life-threatening clots. VLX-1005 is the first and only potent, highly selective inhibitor of human 12-LOX in clinical testing. This asset carries both FDA Orphan Status and Fast Track designations, which are intended to expedite the development of therapies for rare and serious conditions. During the January 2026, J.P. Morgan Healthcare Conference, management highlighted the high incidence of thrombotic complications in HIT, underscoring the urgent need for a therapeutic solution like VLX-1005. Expanding with Factor XIa Inhibition:
The company also added frunexian and EP-7327 to its portfolio in September 2025. Frunexian is a Phase 2-ready intravenous Factor XIa inhibitor designed for acute procedural scenarios, such as bypass surgery or mechanical device support where contact activation triggers clotting. Factor XIa inhibition is a closely watched mechanism because it may reduce clotting risk while limiting bleeding, a major hurdle for existing anticoagulants. Detailed Program Breakdown:
To appreciate the scale of what (CVKD) is building, one must look at the specific indications and the science driving each program. Tecarfarin (Phase 3-Ready): - Mechanism: Next-generation oral vitamin K antagonist.
- Indication: Targeted at patients with ESKD and AFib, and those with Left Ventricular Assist Devices (LVADs).
- Status: Manufacturing has been completed under current good manufacturing practices (cGMP).
- Differentiator: It bypasses the CYP450 metabolism pathway, reducing the risk of drug interactions and providing a more stable therapeutic window.
Frunexian (Phase 2-Ready): - Mechanism: IV Factor XIa inhibitor.
- Indication: Coronary Artery Bypass Grafting (CABG) and other device-driven contact activation scenarios.
- Evidence: Phase 1 studies demonstrated high potency and rapid clearance, making it ideally suited for the acute care environment.
- Potential: Aims to eliminate the "bleeding penalty" associated with traditional anticoagulants in surgical settings.

VLX-1005 (Phase 2): - Mechanism: 12-LOX inhibitor.
- Indication: Heparin-Induced Thrombocytopenia (HIT).
- Significance: HIT remains a life-threatening condition with a high risk of thrombosis despite available therapies.
- Regulatory: Holds FDA and EMA Orphan designations plus FDA Fast Track status.
And this is where the story starts to shift from "interesting pipeline" to "company people are suddenly paying attention to." (CVKD) isn't just assembling programs on paper—it's stacking clinical and operational signals that the market tends to notice when they show up together. When a little-known company, like (CVKD), begins pairing late-stage readiness with fresh assets in high-need settings, visibility can change fast. Strategic Milestone: The J.P. Morgan Healthcare Conference
The presentation at the J.P. Morgan Healthcare Conference on January 12, 2026, was a pivotal moment for the company's visibility. Management used this stage to emphasize that HIT is not just a rare condition but a high-incidence complication that lacks a "gold standard" treatment. By positioning VLX-1005 as the first and only selective 12-LOX inhibitor, (CVKD is effectively claiming leadership in a niche but highly critical segment of hospital-based medicine. Furthermore, the company has emphasized its manufacturing progress. Completing cGMP manufacturing for tecarfarin is a major hurdle cleared. This move signals that (CVKD is not just a research house but an operationally ready late-stage clinical developer capable of moving into pivotal trials. Targeted Response System
As noted in recent updates, the CVKD pipeline is beginning to look less like a typical biotech and more like an "emergency response system" for the cardiovascular ward. Whether it is a patient suffering from HIT, a patient undergoing a CABG procedure, or an ESKD patient needing long-term protection from AFib, CVKD is developing specific tools for specific crises. This "layered approach" is something few emerging companies can match, as most focus on a single mechanism or a single indication. 7 Reasons Why (CVKD) Will Be Topping Our Watchlist Tomorrow Morning —Monday, March 02, 2026
1. Small Float: With fewer than 2M shares available to the public, (CVKD)'s small float could witness big moves if demand begins to shift. 2. Recent Momentum: In just 10 days, (CVKD) made an approximate 73% move, from around $4.91 on Feb. 17 to $8.51 on Feb. 27, 2026. 3. Analyst Targets: Coverage from Noble Capital Markets and Zacks places targets around $45 and $25 on (CVKD), which suggests between 200% to over 450% upside potential from recent ranges. 4. Fresh Data: Late-February updates showed (CVKD) reporting Phase 2 results in HIT with a >25% absolute reduction in thrombotic events versus placebo. 5. Pipeline Depth: Following recent acquisitions, (CVKD) now advances three late-stage clinical programs across chronic, acute, and immune-mediated clotting risk. 6. Regulatory Edge: Through VLX-1005, (CVKD) holds FDA Orphan and Fast Track designations in HIT, a serious condition lacking a gold standard treatment. 7. Operational Progress: With cGMP manufacturing completed for tecarfarin, (CVKD) has cleared a key milestone supporting late-stage clinical advancement. Pull Up (CVKD) Before Tomorrow Morning…
When you step back and look at the full picture, (CVKD) is checking multiple boxes at once. A float of fewer than 2M shares creates a structure where shifts in attention can have an outsized impact, and we've already seen what that can look like with an approximate 73% move in just 10 days from $4.91 to $8.51. Add in analyst targets around $25 and $45—suggesting between 200% and over 450% upside potential from recent ranges—and it's clear why research desks are paying attention. Beyond the tape, the story is being reinforced by substance. Late-February Phase 2 data in HIT showed a >25% absolute reduction in thrombotic events versus placebo, while recent acquisitions expanded (CVKD) into a three-program, late-stage platform spanning chronic, acute, and immune-mediated clotting risk. With FDA Orphan and Fast Track designations for VLX-1005 and cGMP manufacturing completed for tecarfarin, (CVKD) is pairing clinical progress with operational execution. We will have all eyes on (CVKD) tomorrow morning—Monday, March 02, 2026. Take a look at (CVKD) before you call it a night. Also, keep a lookout for my morning update, it could be hitting early. Have a good night. Sincerely, Gary Silver
Managing Editor,
Market Crux
| 




Tidak ada komentar:
Posting Komentar