Any content you receive is for information purposes only. Always conduct your own research.*Sponsored
Market Crux Has All Eyes on Alzamend Neuro, Inc. (Nasdaq: ALZN) This Morning—Thursday, March 26, 2026
Don't Miss The Next Breakout—Get Real-Time Alerts Sent Directly
To Your Phone. Up To 10X Faster Than Email.
Full Coverage On (ALZN) Is Starting Now
Put (ALZN) On Your Radar While It's Still Early…
March 26, 2026
Radar Lighting Up | (ALZN) Showcases Approx. 29% Move on Breaking News Dear Reader, Market Crux is following breaking news this morning involving Alzamend Neuro, Inc. (Nasdaq: ALZN), with the market already starting to respond to the latest development. Earlier today, (ALZN) tapped $2.33, marking an approximate 29% overnight move from yesterday's $1.80 range, as the company reported positive Phase II topline data from its "Lithium in Brain" clinical study. The results showed that AL001 achieved bioequivalence to standard lithium carbonate in blood levels, while also demonstrating numerically higher lithium concentrations across all measured brain regions, including the whole brain. The study further indicated faster brain uptake, reaching peak levels sooner than traditional lithium. The trial, conducted in collaboration with Massachusetts General Hospital, also utilized advanced MRI and spectroscopy imaging to directly measure lithium distribution in the brain—marking a key step forward in evaluating the program. With clinical data now in hand, a second Phase II study already underway, and additional trials planned across multiple neurological conditions, this development has quickly placed (ALZN) in focus. We're keeping all eyes on (ALZN) as this story continues to unfold. If you missed my earlier email coverage, keep reading to quickly get up to speed on why we're so excited to be highlighting (ALZN) today. ===== Market Crux is tracking a biotech story that appears to be gaining momentum heading into a potentially important catalyst. Alzamend Neuro, Inc. (Nasdaq: ALZN) could start to draw increased attention as its end-of-March data readout approaches and interest builds around its re-engineered lithium program. That's one of the key reasons why (ALZN) just hit the Market Crux radar and is stopping our watchlist this morning—Thursday, March 26, 2026. But keep in mind, (ALZN) has less than 4M shares listed as available to the public, according to MarketWatch. When companies have small floats like this, the potential exists for big moves if demand begins to shift. In fact, over the last month, (ALZN) has already made an approximate 48% move from around $1.58 on February 17 to $2.35 on March 16, according to data available from Barchart. What's even more interesting, Analyst Edward Woo, CFA, of Ascendiant Capital Market, recently highlighted a $28 target for (ALZN), which suggests over 1,400% upside potential from this week's $1.80 range. 
(ALZN) is at the center of that effort, developing a re-engineered version of lithium designed to reach the brain more effectively while limiting toxic side effects elsewhere in the body. As the company approaches this important milestone, the biotech world is watching for results that could reshape the standard of care for a vast number of patients living with Alzheimer's and psychiatric disorders. The Mission to Modernize Neuroscience
Alzamend Neuro, Inc. (Nasdaq: ALZN) is a clinical-stage biopharmaceutical company dedicated to developing novel products that address the massive unmet needs in the treatment of Alzheimer's disease, bipolar disorder, major depressive disorder (MDD), and post-traumatic stress disorder (PTSD). Unlike many biotechnology firms that chase entirely unproven biological targets, (ALZN) is utilizing a "known-quantity" approach by taking lithium—a mineral with decades of proven efficacy—and optimizing its delivery through cutting-edge chemical engineering. The company's strategy is built on the reality that while lithium is one of the most effective tools in a psychiatrist's arsenal, its "old-school" formulations are fraught with complications. Current lithium salts, like lithium carbonate, often struggle to cross the blood-brain barrier efficiently. This forces doctors to prescribe higher doses, which leads to high concentrations in the bloodstream. These systemic levels can be detrimental to the kidneys and thyroid, requiring patients to undergo lifelong, invasive blood monitoring. (ALZN) intends to break this cycle of toxicity with its primary candidate, AL001. Pipeline Overview
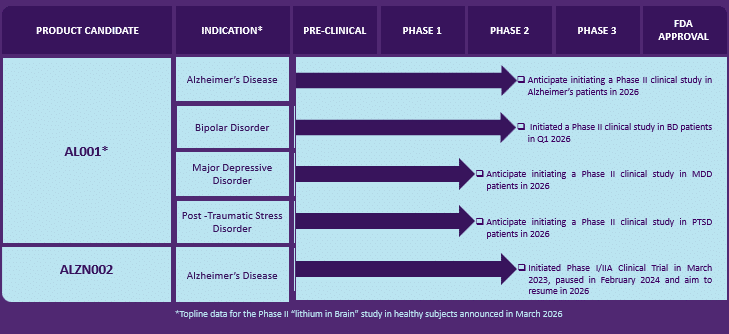
Alzamend Neuro, Inc. (Nasdaq: ALZN)'s research program currently includes two primary therapeutic candidates focused on neurological and psychiatric conditions. (ALZN)'s lead program, AL001, is a patented ionic cocrystal formulation of lithium designed to improve how lithium reaches the brain and is being studied for Alzheimer's disease, bipolar disorder, major depressive disorder, and PTSD. In addition to AL001, (ALZN) is also developing ALZN002, a cell-based immunotherapy approach designed to stimulate the immune system to help combat Alzheimer's disease by targeting beta-amyloid buildup in the brain. See the company's latest corporate presentation here. AL001: The Ionic Cocrystal Breakthrough
At the heart of the Alzamend Neuro story is AL001, a patented ionic cocrystal of lithium, salicylate, and L-proline. This is not just a new pill; it is a sophisticated delivery vehicle designed to improve the pharmacokinetics of lithium. By combining these specific compounds, the AL001 formulation is designed to improve how lithium reaches the brain. In preclinical mouse studies, this technology demonstrated a remarkable ability to achieve higher brain uptake while maintaining lower levels in the blood compared to standard lithium carbonate. This "brain-first" approach is particularly vital for the company's target indications. For an Alzheimer's patient, the goal is neuroprotection and the reduction of cognitive decline; for a patient with bipolar disorder, it is mood stabilization. Both require the drug to reach the central nervous system. By potentially achieving therapeutic brain exposure with a lower systemic dose, (ALZN) aims to offer the benefits of lithium without the constant threat of organ damage. The "Lithium in Brain" Phase II Potential Catalyst

Recent developments have added another layer to the story. The company announced it has initiated a Phase II clinical trial of AL001 in patients with bipolar disorder type I, being conducted in collaboration with Massachusetts General Hospital. The study is designed to compare AL001 directly against marketed lithium carbonate, measuring lithium levels in both the blood and specific brain structures using advanced MRI and spectroscopy imaging technology. This newly initiated trial builds on the company's previously completed "Lithium in Brain" imaging study in healthy subjects, with topline data from that study expected by the end of March 2026—which could be any day now. The most anticipated milestone for Alzamend Neuro is the upcoming topline data from its Phase II "Lithium in Brain" study, conducted in partnership with Massachusetts General Hospital. This is not a standard clinical trial; it utilizes state-of-the-art 7Li MRI technology equipped with a custom-built head coil. This advanced imaging allows researchers to literally map lithium inside the human brain in real-time. The MRI and MRS neuroimaging methods were developed by the lab of Dr. Ovidiu Andronesi, the study's principal investigator, Associate Professor of Radiology at Harvard University and the Director of Multinuclear Metabolic Imaging, Martinos Center for Biomedical Imaging, Department of Radiology, MGH, Harvard Medical School. The study compares AL001 head-to-head with a marketed lithium carbonate product in healthy subjects. The goal is to determine whether (ALZN)'s formulation can deliver more lithium to specific brain structures while maintaining safer blood levels compared with lithium carbonate. The clinical portion of this trial was completed in late 2025, and the topline data is expected in the first quarter of 2026. This data will provide a foundational step toward demonstrating proof‑of‑concept for the AL001 platform. The company is aiming for the topline human data to mirror the preclinical proof‑of‑concept results in mice, which demonstrated reduced systemic exposure and enhanced brain penetration in target areas. Removing Commercial Barriers: The TDM Factor
One of the greatest barriers to lithium adoption is therapeutic drug monitoring (TDM). Because the line between a therapeutic dose and a toxic dose is so thin with traditional lithium, patients must have their blood drawn frequently. This is a massive inconvenience for patients and a significant liability for physicians. (ALZN) believes that because AL001 can maintain safer systemic levels, it may eliminate the need for TDM. If the FDA eventually approves a lithium treatment that does not require constant blood tests, the market potential could expand exponentially. It would allow lithium to be prescribed more freely in primary care settings and for fragile populations, such as the elderly, who often have compromised kidney function and cannot risk standard lithium therapy. Market Potential and Strategic Positioning
The commercial landscape for AL001 is broad. Alzheimer's disease alone remains one of the largest unmet-need markets in global medicine, where even therapies with modest cognitive benefits can generate significant annual sales. By targeting four major indications—Alzheimer's, Bipolar, MDD, and PTSD—Alzamend Neuro is positioning AL001 as a platform-level asset.
The company is led by an experienced management team and guided by a Scientific Advisory Board that includes world-renowned experts in neuroscience. This leadership has been instrumental in securing partnerships with top-tier institutions like Massachusetts General Hospital and ensuring that the clinical program is designed to meet the highest regulatory standards. 
7 Factors Putting (ALZN) At The Top Of This Morning's Watchlist —Thursday, March 26, 2026.…
1. Analyst Target: Ascendiant Capital Market analyst Edward Woo, CFA, recently highlighted a $28 target on (ALZN), which suggests over 1,400% upside potential from its recent $1.80 range. 2. Low Float: With fewer than 4M shares available to the public according to MarketWatch, (ALZN)'s small float could have the potential to witness big moves if demand begins to shift. 3. Recent Momentum: Over the past month, (ALZN) moved approximately 48%, moving from around $1.58 to $2.35 based on Barchart data. 4. Advanced Imaging: Researchers evaluating (ALZN) are using specialized 7Li MRI mapping technology that can measure lithium distribution inside the human brain in real time. 5. Novel Formulation: The patented ionic cocrystal used by (ALZN) combines lithium, salicylate, and L-proline in a formulation designed to improve how lithium reaches the brain. 6. Large Indications: The development program surrounding (ALZN) is focused on Alzheimer's disease, bipolar disorder, major depressive disorder, and PTSD—four major neurological and psychiatric conditions with substantial global patient populations, including over 40M in the United States. 7. Upcoming Data: Topline results from a completed imaging study are expected by the end of March, putting (ALZN) in focus in the near term. Put (ALZN) On Your Radar While It's Still Early…
Taken together, several factors have put (ALZN) on our radar right now. With fewer than 4M shares available to the public, a recent 48% move (approx.) over the past month, and outside coverage highlighting a $28 target—which suggests over 1,400% upside potential from its recent $1.80 range—the name has started to stand out. At the same time, the company is advancing a novel lithium formulation supported by advanced brain imaging techniques, while targeting multiple large neurological and psychiatric conditions. With topline data from a completed imaging study expected by the end of March, the near-term timeline is adding another layer of focus. (ALZN) is front and center for us this morning—Thursday, March 26, 2026. Go ahead and pull up (ALZN) while it's still early. Also, be on the lookout for my next update—it could be landing in your inbox very shortly. Sincerely, Gary Silver
Managing Editor, Market Crux
| 




Tidak ada komentar:
Posting Komentar